 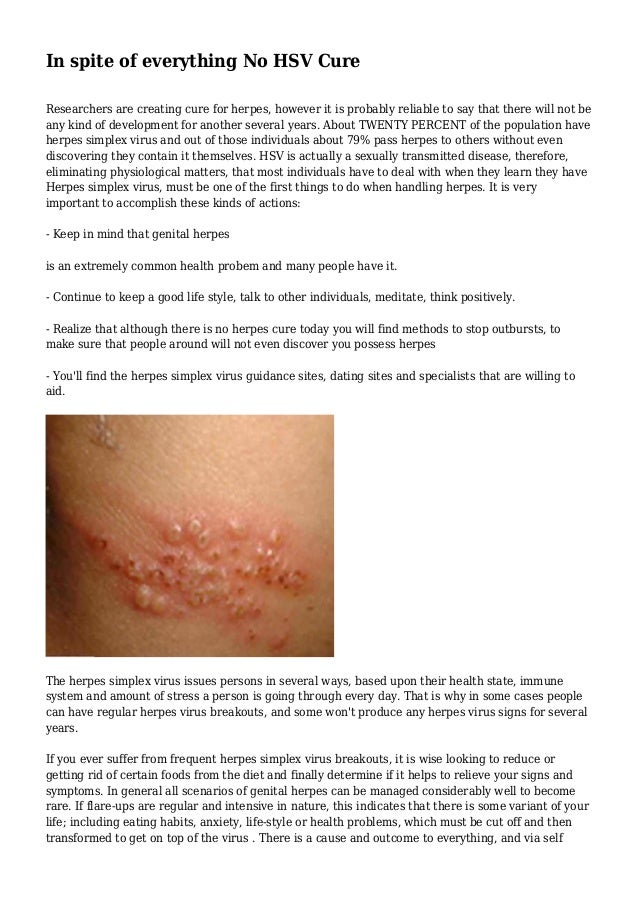
The doctor also urged people who have recovered from Covid-19 to remain cautious as they can attract such diseases due to their weakened immune system post hospitalisation. He also underlined the heavy cost for the treatment, although the patient is being supervised carefully due to the availability of medicines. Dr Tyagi termed the virus “very dangerous", and said if there is a delay in treatment then this virus can be deadlier than Covid-19.ĭr Tyagi also informed that the patient is undergoing treatment at his hospital. 
The treatment of this disease is very costly, and the emergence of this virus has alerted the scientific community as it is considered deadly.ĭr BP Tyagi from Ghaziabad said the first-ever case of herpes simplex virus in India was found in the nose of a patient. Now, a hospital in Ghaziabad, just a few kilometres from the national capital Delhi, has reported the first case of herpes simplex virus infection, and has even termed the new disease “very dangerous".Īccording to doctors, Covid recovered patients who have weakened immunity or suffering from an existing ailment are the most susceptible to herpes simplex virus. According to confirmed reports, scientists from China will soon start testing the HIV vaccine on 160 human volunteers. Then cases of white and yellow fungus infections also emerged. While no cure for the viral disease has been found till date, in what may be termed promising, human trials may soon start, with a vaccine developed by a group of Chinese scientists having received a green signal. As the second wave was peaking, some recovered Covid patients were diagnosed with black fungus. Scientists found that a new deadly variant of coronavirus B.1.617.2 was driving the explosive surge in cases and deaths. Due to EU data protection law this shall not be accompanied by personal data.The second wave of Covid-19 has given many shocks to the country. 
When providing medical information about the patient, please note that only anonymized data shall be included in any patient information. Physician should contact the myTomorrows medical team for more information on this EAP. If the physician agrees to a treatment with Pritelivir oral tablets and to oversee the patient's treatment, the To get access, patients need to consult with their treating physician to explore all treatment options. Studies from January 2001 and September 2001 have found penciclovir to be effective as a treatment. There are two types of herpes simplex virus. It primarily spreads by skin-to-skin contact. Eligible patients for an EAP are in high medical need, cannot participate in a Clinical Trial, and have exhausted all registered treatment options. Herpes simplex virus (HSV), known as herpes, is a common infection that can cause painful blisters or ulcers. EAPs (also known as “Expanded Access Program” or “Named-Patient Use”) offer an ethical, compliant, and controlled way of providing treatment with a medicine currently not licensed in a patient’s country of residence. AiCuris also supports expanded access requests for Pritelivir oral tablets for the treatment of eligible patients and collaborates with myTomorrows to facilitate early access to Pritelivir for acyclovir-resistant mucocutaneous HSV infections in immunocompromised patients. A phase 2/3 trial in immunocompromised patients is ongoing. Please visit (study identifier: NCT03073967) to see whether your patient meets trial criteria and to find contact details of a potential trial site. It has received the US FDA Breakthrough Therapy Designation.Įarly Access to Pritelivir oral tablets for immunocompromised patients with resistant HSV infectionsĪiCuris develops Pritelivir oral tablets for the treatment of acyclovir-resistant mucocutaneous herpes simplex virus (HSV) infections. The development of Pritelivir for patients with high medical need is in clinical Phase 3 and Pritelivir is not yet available as a marketed product. Pritelivir is being developed for the treatment of acyclovir-resistant and dual-resistant (resistant to acyclovir and intolerant or resistant to foscarnet) infections in immune-compromised patients. In contrast to currently used nucleoside analogues, Pritelivir does not require activation by viral enzymes and can thus protect uninfected cells. It belongs to a new chemical class and acts via a novel mechanism of action (inhibition of the viral helicase-primase enzyme complex). Pritelivir is a highly potent inhibitor of HSV replication. Herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) infections lead to lifelong persistence of the virus, with frequent and sometimes painful recurrences, and carry the risk of serious complications in immune-compromised patients. Treating acyclovir-resistant HSV infection 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
